
Breaking News
 The Real Cost of Supporting the Zionist Secular State of Israel
The Real Cost of Supporting the Zionist Secular State of Israel
 Treasury Yield 30 Years (^TYX)
Treasury Yield 30 Years (^TYX)
 Apoplectic Netanyahu rages at Trump in private as 'disastrous' Iran deal leaves him longing.
Apoplectic Netanyahu rages at Trump in private as 'disastrous' Iran deal leaves him longing.
 The American Consumer Is Piss Broke
The American Consumer Is Piss Broke
Top Tech News
 Elon and SpaceX Have Made AI Training 10 Times Faster
Elon and SpaceX Have Made AI Training 10 Times Faster
 Oklo COO Says Nuclear Waste Could Power America For 150 Years
Oklo COO Says Nuclear Waste Could Power America For 150 Years
 SpaceX Announces LARGEST Starship Mission Ever! They've never done this before!
SpaceX Announces LARGEST Starship Mission Ever! They've never done this before!
 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...
 Our Emergency Water Plan Wasn't Good Enough - So We Built This
Our Emergency Water Plan Wasn't Good Enough - So We Built This
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
AstraZeneca Coronavirus Vaccine to Be Pulled Worldwide for 'Commercial' Reasons
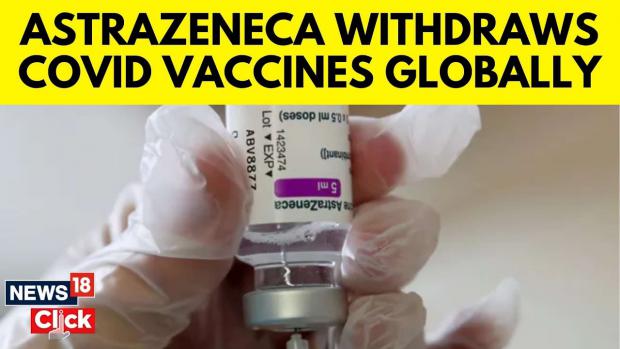
The Oxford-AstraZeneca coronavirus vaccine will be withdrawn worldwide just months after the firm admitted that the jab causes a rare but dangerous side effect, however, AstraZeneca insists that the decision to pull the vaccine was purely made for commercial reasons.
AstraZeneca pulled its "marketing authorisation" for its COVID-19 vaccine, known commercially as Vaxzevria, in the European Union on Tuesday and plans to do so in the UK and other countries where it was deployed in the coming months. The decision will not impact the United States, where the jab never received approval to go to market.
The pharmaceutical giant insisted that the decision was made for commercial considerations, saying that the vaccine is no longer being manufactured and that more recent vaccines have been developed to confront novel variants of the Chinese virus.



