
Breaking News
 "The World Is Losing Trust": Foreign Investment In Germany Plunges To Lowest Level Since 2
"The World Is Losing Trust": Foreign Investment In Germany Plunges To Lowest Level Since 2
 The Race To Build The World's Tallest Skyscraper
The Race To Build The World's Tallest Skyscraper
 At Last Minute, SEC Suddenly Delays Plan To Allow Crypto Versions Of US Stocks
At Last Minute, SEC Suddenly Delays Plan To Allow Crypto Versions Of US Stocks
 Debt Remembered And Debt Ignored
Debt Remembered And Debt Ignored
Top Tech News
 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...
 Our Emergency Water Plan Wasn't Good Enough - So We Built This
Our Emergency Water Plan Wasn't Good Enough - So We Built This
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Thermal Energy Stored for Month in New Material
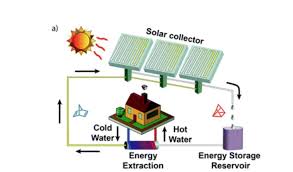
The amount of solar power in winter drops by 90%. Solar energy could provide a larger share of global power if there was a way to store all of the worlds energy usage for months. The development of solar energy can potentially meet the growing requirements for a global energy system beyond fossil fuels, but necessitates new scalable technologies for solar energy storage. Thermal energy can be used for a broad range of applications such as domestic heating, industrial process heating and in thermal power processes. One promising way to store solar thermal energy is so-called molecular solar thermal (MOST) energy storage systems, where a photoswitchable molecule absorbs sunlight and undergoes a chemical isomerization to a metastable high energy species. Here we present an optimized MOST system (providing a high energy density of up to 0.4 MJ kg−1), which can store solar energy for a month at room temperature and release the thermochemical energy "on demand" in a closed energy storage cycle. In addition to a full photophysical characterization, solar energy capture of the present system is experimentally demonstrated by flowing the MOST system through an outdoor solar collector (≈900 cm2 irradiated area). Moreover, catalyst systems were identified and integrated into an energy extraction device leading to high temperature gradients of up to 63 °C (83 °C measured temperature) with a short temperature ramp time of only a few minutes. The underlying step-by-step mechanism of the catalytic reaction is modelled in detail using quantum chemistry calculations, successfully rationalizing the experimental observations.



