
Breaking News
 Parsley: Nature's Powerful Ally in the Fight Against Cancer and Chronic Disease
Parsley: Nature's Powerful Ally in the Fight Against Cancer and Chronic Disease
 Iran's Khamenei Killed & Austin Mass Shooting | PBD #750
Iran's Khamenei Killed & Austin Mass Shooting | PBD #750
 'Just Flatten It' Is the West's Answer to War
'Just Flatten It' Is the West's Answer to War
Top Tech News
 US particle accelerators turn nuclear waste into electricity, cut radioactive life by 99.7%
US particle accelerators turn nuclear waste into electricity, cut radioactive life by 99.7%
 Blast Them: A Rutgers Scientist Uses Lasers to Kill Weeds
Blast Them: A Rutgers Scientist Uses Lasers to Kill Weeds
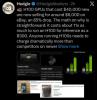 H100 GPUs that cost $40,000 new are now selling for around $6,000 on eBay, an 85% drop.
H100 GPUs that cost $40,000 new are now selling for around $6,000 on eBay, an 85% drop.
 We finally know exactly why spider silk is stronger than steel.
We finally know exactly why spider silk is stronger than steel.
 She ran out of options at 12. Then her own cells came back to save her.
She ran out of options at 12. Then her own cells came back to save her.
 A cardiovascular revolution is silently unfolding in cardiac intervention labs.
A cardiovascular revolution is silently unfolding in cardiac intervention labs.
 DARPA chooses two to develop insect-size robots for complex jobs like disaster relief...
DARPA chooses two to develop insect-size robots for complex jobs like disaster relief...
 Multimaterial 3D printer builds fully functional electric motor from scratch in hours
Multimaterial 3D printer builds fully functional electric motor from scratch in hours
 WindRunner: The largest cargo aircraft ever to be built, capable of carrying six Chinooks
WindRunner: The largest cargo aircraft ever to be built, capable of carrying six Chinooks
First totally synthetic human brain model has been realized
This environment is carefully structured and carries signals that guide how cells behave and interact.
3D tissue-engineered models have strong potential to mimic the brain's complex structure and function. Yet it's still difficult to reproduce the brain's subtle design features in lab settings, since current methods often miss the fine details that shape cell behavior.
Scientists at the University of California, Riverside have now, for the first time, developed functional brain-like tissue without relying on animal-derived materials or biological coatings. Their innovation, called the Bijel-Integrated PORous Engineered System (BIPORES), offers a new, fully synthetic platform for neural tissue engineering.
This breakthrough could significantly reduce, and potentially eliminate, the need to use animal brains in research. It also supports the US FDA's ongoing initiative to phase out animal testing in drug development.
The new material is mainly made of polyethylene glycol (PEG), a chemically neutral polymer. On its own, PEG is like Teflon to cells; they slide right off. Usually, it needs a helping hand from proteins like laminin or fibrin to keep cells from falling off.
Scientists previously developed a technique called STrIPS to continuously produce tiny particles, fibers, and films with sponge-like internal structures. However, until now, these materials could only be made up to about 200 micrometers thick, limited by how molecules move through the material during formation.
To overcome this, researchers developed the BIPORES system. It combines large-scale fibrous shapes with intricate pore patterns inspired by bicontinuous interfacially jammed emulsion gels (bijels), soft materials with smooth, saddle-shaped internal surfaces. These BIPORES fibers are made from a gel-like PEG solution, which is transformed into a porous network and stabilized using silica nanoparticles.
Using a custom microfluidic setup and a bioprinter, the team then built 3D structures with layered, interconnected pores. These allow nutrients and waste to move freely and support deep cell growth. When tested with neural stem cells, the material encouraged strong cell attachment, growth, and even the formation of active nerve connections.
"Since the engineered scaffold is stable, it permits longer-term studies," said Prince David Okoro, the study's lead author. "That's especially important as mature brain cells are more reflective of real tissue function when investigating relevant diseases or traumas."
To build the scaffold, the team used a special liquid mix made of PEG, ethanol, and water. PEG doesn't mix well with water, so it behaves like oil, while ethanol helps everything blend smoothly. This mix flowed through tiny glass tubes.

 March 2: nobody to call
March 2: nobody to call
 RNA Crop Spray: Should We Be Worried?
RNA Crop Spray: Should We Be Worried?

