
Breaking News
 Tina Peters, granted clemency by Colorado's Dem governor
Tina Peters, granted clemency by Colorado's Dem governor
 Tulsi Gabbard at center of explosive CIA claim as JFK and MKUltra files 'vanish from her office*
Tulsi Gabbard at center of explosive CIA claim as JFK and MKUltra files 'vanish from her office*
 Bessent Says US, China To Launch AI Safety Talks After Trump-Xi Meeting In Beijing
Bessent Says US, China To Launch AI Safety Talks After Trump-Xi Meeting In Beijing
 Cuba Depletes Fuel As Blackouts Worsen, Putting Havana's Communists Under Pressure...
Cuba Depletes Fuel As Blackouts Worsen, Putting Havana's Communists Under Pressure...
Top Tech News
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
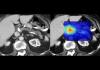 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
A multi-terrain robot from China is going viral, not because of raw speed or power...
 The World's Biggest Fusion Reactor Just Hit A Milestone
The World's Biggest Fusion Reactor Just Hit A Milestone
 Wow. Researchers just built an AI that can control your body...
Wow. Researchers just built an AI that can control your body...
 Google Chrome silently installs a 4 GB AI model on your device without consent
Google Chrome silently installs a 4 GB AI model on your device without consent
 The $5 Battery That Never Dies - Edison Buried This 100 Years Ago
The $5 Battery That Never Dies - Edison Buried This 100 Years Ago
Experimental cancer drug that targets common genetic fault can shrink tumours...

An experimental cancer drug that targets a common genetic fault has been shown to shrink tumours by up to 67 per cent in just six weeks.
Scientists tested their newly developed anti-cancer drug AMG 510 on four patients, two of whom saw their tumours shrink. It did not work for the others.
In studies on mice, the treatment helped to shrink, and in some cases eradicate, the growing tumours.
The daily pill works by turning 'off' the KRAS gene. Mutated forms of the genes are permanently trapped 'on', causing cancerous cells to multiply.
This abnormal activity fuels the development of up to 50 per cent of lung cancers, as well as some pancreatic and bowel cancers.
The pharmaceutical company described their findings as a 'milestone' for patients with cancer KRAS-mutant cancers.
The research was led by by pharmaceutical company Amgen Research, based in Thousand Oaks, California.
The findings, published in Nature, are the first to report the effects of inhibiting KRAS with a drug.



