
Breaking News
 Economist Issues a Chilling Warning (You Should Prepare)
Economist Issues a Chilling Warning (You Should Prepare)
 The Internet Is Getting Harder to Trust | Josh Smith From #485 | The Way I Heard It
The Internet Is Getting Harder to Trust | Josh Smith From #485 | The Way I Heard It
 DIY LFP Battery Explosion! Is it Possible??
DIY LFP Battery Explosion! Is it Possible??
 House Leadership Delays Vote on Ending the Iran War
House Leadership Delays Vote on Ending the Iran War
Top Tech News
 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...
 Our Emergency Water Plan Wasn't Good Enough - So We Built This
Our Emergency Water Plan Wasn't Good Enough - So We Built This
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
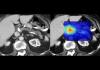 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
New Materials Could Turn Water into the Fuel of the Future and puts chemical ...

Scientists at the Department of Energy's Lawrence Berkeley National Laboratory (Berkeley Lab) and the California Institute of Technology (Caltech) have—in just two years—nearly doubled the number of materials known to have potential for use in solar fuels.
They did so by developing a process that promises to speed the discovery of commercially viable generation of solar fuels that could replace coal, oil, and other fossil fuels.
Solar fuels, a dream of clean-energy research, are created using only sunlight, water, and carbon dioxide. Researchers are exploring a range of possible target fuels, but one possibility is to produce hydrogen by splitting water.
Each water molecule is comprised of an oxygen atom and two hydrogen atoms. Pure hydrogen is highly flammable, making it an ideal fuel. If you could find a way to extract that hydrogen from water using sunlight, then, you would have a plentiful and renewable energy source. The problem, however, is that water molecules do not simply break down when sunlight shines on them—if they did, the oceans would not cover three-fourths of the planet. Instead, they need a little help from a solar-powered catalyst.
To create practical solar fuels, scientists have been trying to develop low-cost and efficient materials that perform the necessary chemistry using only visible light as an energy source.
Over the past four decades, researchers identified only 16 of these "photoanode" materials. Now, using a new high-throughput method of identifying new materials, a team of researchers led by Caltech's John Gregoire and Berkeley Lab's Jeffrey Neaton, Kristin Persson, and Qimin Yan have found 12 promising new photoanodes.




