
Breaking News
 How to Make Infinite FREE Chicken Feed
How to Make Infinite FREE Chicken Feed
 Amish's 25 BANNED Epsom Salt Hacks from the 1950s That Doctors Now Say Were GENIUS
Amish's 25 BANNED Epsom Salt Hacks from the 1950s That Doctors Now Say Were GENIUS
 The Korean Bucket That Grows Unlimited Plant Steroids -- Doubles Every 48 Hours
The Korean Bucket That Grows Unlimited Plant Steroids -- Doubles Every 48 Hours
 This CHICKEN Lays 365 EGGS Per Year. Never Gets Sick. Why Did They Make It Disappear?
This CHICKEN Lays 365 EGGS Per Year. Never Gets Sick. Why Did They Make It Disappear?
Top Tech News
 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...
 Our Emergency Water Plan Wasn't Good Enough - So We Built This
Our Emergency Water Plan Wasn't Good Enough - So We Built This
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
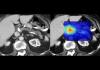 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Ultra-high density hydrogen storage holds twice as much as liquid H2

Hydrogen is finding plenty of applications as a clean fuel – in trucking and commercial vehicles, short range aviation and shipping, for example, where it carries considerably more energy per weight and volume than lithium batteries and can deliver superior range figures and quick refueling. You can burn it more or less like gasoline, or run it through a fuel cell to generate electric power.
It has the highest energy per mass of any fuel, but it's a pain to store. Keep it in gas tanks and you'll need some 700 atmospheres' worth of compression. Keep it as a liquid, and you'll need to maintain cryogenic temperatures just 20 degrees above absolute zero. And even when squashed into a supercooled liquid, it might be lightweight, but it takes up a surprising and inconvenient amount of volume, making it both energy-hungry and tough to package where space is an issue.
Now, Korean researchers say they've created a material that stores hydrogen at double the density of its cryogenic liquid form. "Our innovative material represents a paradigm shift in the realm of hydrogen storage, offering a compelling alternative to traditional approaches," said Hyunchul Oh, from the Ulsan National Institute of Science and Technology (UNIST), lead author on this new research.
As a molecule, hydrogen can physically adsorb into a porous material in a process called physisorption. Highly porous materials have previously demonstrated the ability to store a large amount of hydrogen per unit mass, but they've struggled to store a lot of energy within a small volume.
Until now. The team synthesized nanoporous magnesium borohydride (Mg(BH4)2), a framework with partially negatively-charged hydrogen atoms forming the nanopore's inner surface, enabling the uptake of hydrogen and nitrogen. Although both nitrogen and hydrogen can enter the pores, the researchers found that the gas uptake for hydrogen was larger by a factor of three as both occupy different adsorption sites in the pores.



