
Breaking News
 Gorka Advocates Violating the First Amendment for Israel
Gorka Advocates Violating the First Amendment for Israel
 Palantir Manifesto Shows The Clear Convergence Of Technofascism With Technocracy
Palantir Manifesto Shows The Clear Convergence Of Technofascism With Technocracy
 Washington's Democrat ex-governor says she's disgusted at millionaires' tax...
Washington's Democrat ex-governor says she's disgusted at millionaires' tax...
 The Odyssey Backlash Goes NUCLEAR - WTF Nolan?
The Odyssey Backlash Goes NUCLEAR - WTF Nolan?
Top Tech News
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
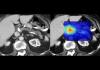 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
A multi-terrain robot from China is going viral, not because of raw speed or power...
 The World's Biggest Fusion Reactor Just Hit A Milestone
The World's Biggest Fusion Reactor Just Hit A Milestone
 Wow. Researchers just built an AI that can control your body...
Wow. Researchers just built an AI that can control your body...
 Google Chrome silently installs a 4 GB AI model on your device without consent
Google Chrome silently installs a 4 GB AI model on your device without consent
 The $5 Battery That Never Dies - Edison Buried This 100 Years Ago
The $5 Battery That Never Dies - Edison Buried This 100 Years Ago
Pouch of stem cells implanted in trial to cure type 1 diabetes
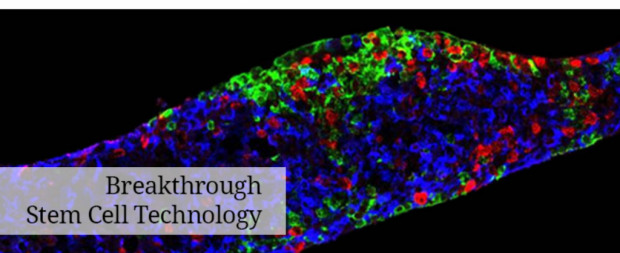
The first implant procedures of the clinical trial took place at the University of Alberta Hospital in Edmonton, Alberta, and the UC San Diego School of Medicine's Altman Clinical Trials Research Institute. The goal of the open-label clinical trial is to evaluate the PEC-Direct product candidate for safety and definitive evidence of efficacy. In the coming months, the company expects to expand the trial to additional centers including the University of Minnesota and other sites in the US and Canada.
The first cohort of type 1 diabetes patients is receiving multiple small-format cell-filled devices called sentinels in order to evaluate safety and implant viability. These sentinel units will be removed at specific time points and examined histologically to provide early insight into the progression of engraftment and maturation into pancreatic islet cells including insulin-producing beta cells. A second cohort of up to 40 patients is expected to begin enrolling later this year to evaluate both safety and efficacy. The primary efficacy measurement in the trial will be the clinically relevant production of insulin, as measured by the insulin biomarker C-peptide, in a patient population that has little to no ability to produce endogenous insulin at the time of enrollment. Other important endpoints will be evaluated including injectable insulin usage and the incidence of hypoglycemic events. ViaCyte's goal is to demonstrate early evidence of efficacy in the first half of 2018 and definitive efficacy 6 to 12 months later.



