
Breaking News
 Tulsi Gabbard at center of explosive CIA claim as JFK and MKUltra files 'vanish from her office*
Tulsi Gabbard at center of explosive CIA claim as JFK and MKUltra files 'vanish from her office*
 Bessent Says US, China To Launch AI Safety Talks After Trump-Xi Meeting In Beijing
Bessent Says US, China To Launch AI Safety Talks After Trump-Xi Meeting In Beijing
 Cuba Depletes Fuel As Blackouts Worsen, Putting Havana's Communists Under Pressure...
Cuba Depletes Fuel As Blackouts Worsen, Putting Havana's Communists Under Pressure...
 Russia Sends Over 1500 Missiles, Drones On Ukraine In 48 Hours After V-Day Ceasefire
Russia Sends Over 1500 Missiles, Drones On Ukraine In 48 Hours After V-Day Ceasefire
Top Tech News
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
A multi-terrain robot from China is going viral, not because of raw speed or power...
 The World's Biggest Fusion Reactor Just Hit A Milestone
The World's Biggest Fusion Reactor Just Hit A Milestone
 Wow. Researchers just built an AI that can control your body...
Wow. Researchers just built an AI that can control your body...
 Google Chrome silently installs a 4 GB AI model on your device without consent
Google Chrome silently installs a 4 GB AI model on your device without consent
 The $5 Battery That Never Dies - Edison Buried This 100 Years Ago
The $5 Battery That Never Dies - Edison Buried This 100 Years Ago
39-Year-Old Becomes First US Patient to Receive Innovative Artificial Heart Prosthetic
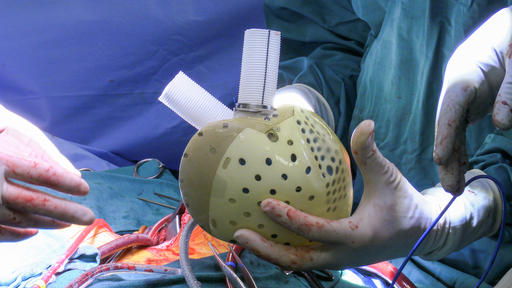
The artificial heart was developed by a French company, CARMAT and has been approved for use and sale in Europe.
Last year, the company received U.S. FDA approval to begin studies and enroll 10 patients with end-stage biventricular heart failure—people who are suffering on the waiting list for a heart donor—and offer a life-saving bridge before transplant.
"We are encouraged that our patient is doing so well after the procedure Monday," said Dr. Carmelo Milano, a transplant surgeon and the principal investigator of the device study at Duke. "As we evaluate this device, we are both excited and hopeful that patients who otherwise have few to no options could have a lifeline."
The North Carolina patient, Matthew Moore, is just 39-years-old and was referred to Duke in June after a sudden, unexpected diagnosis of heart failure. Moore and his wife, Rachel, recently adopted their two-year-old foster son, Marshall, and arrived at Duke expecting only to undergo heart bypass surgery.



