
Breaking News
 How to Make Infinite FREE Chicken Feed
How to Make Infinite FREE Chicken Feed
 Amish's 25 BANNED Epsom Salt Hacks from the 1950s That Doctors Now Say Were GENIUS
Amish's 25 BANNED Epsom Salt Hacks from the 1950s That Doctors Now Say Were GENIUS
 The Korean Bucket That Grows Unlimited Plant Steroids -- Doubles Every 48 Hours
The Korean Bucket That Grows Unlimited Plant Steroids -- Doubles Every 48 Hours
 This CHICKEN Lays 365 EGGS Per Year. Never Gets Sick. Why Did They Make It Disappear?
This CHICKEN Lays 365 EGGS Per Year. Never Gets Sick. Why Did They Make It Disappear?
Top Tech News
 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...
 Our Emergency Water Plan Wasn't Good Enough - So We Built This
Our Emergency Water Plan Wasn't Good Enough - So We Built This
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
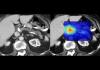 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
New catalyst material can help spur the rise of lithium-sulfur batteries

But LSBs can only be recharged a few times before degrading. Now, South Korean researchers might have just found the right catalyst to improve the lifespan and performance of these batteries. In a study published in the journal ChemSusChem, the researchers described how they used the compound cobalt oxalate to reverse the degradation of LSBs.
Improving the lifespan of lithium-sulfur batteries
LSBs are not yet widely used today because charging them causes a buildup of solid lithium sulfide and liquid lithium polysulfide. In turn, these chemical deposits degrade the sulfur cathode (positively charged electrode) and lithium anode (negatively charged electrode), which effectively reduces the lifespan of the batteries and may even cause them to catch fire.
The researchers then looked for a catalyst – a substance that increases the rate of a chemical reaction without undergoing any permanent chemical change – that can make the degradation of LSBs reversible during use.
"While looking for a new electrocatalyst for the LSBs, we recalled a previous study we had performed with cobalt oxalate in which we had found that negatively charged ions can easily adsorb on this material's surface during electrolysis," said Jaeyoung Lee, a professor of environmental engineering at the Gwangju Institute of Science and Technology in South Korea and one of the study researchers.
This led Lee and his team to hypothesize that cobalt oxalate could have a protective effect against the chemical deposits. To test their hypothesis, they made an LSB and added a layer of cobalt oxalate on the surface of the sulfur cathode.



