
Breaking News
 How to Make Infinite FREE Chicken Feed
How to Make Infinite FREE Chicken Feed
 Amish's 25 BANNED Epsom Salt Hacks from the 1950s That Doctors Now Say Were GENIUS
Amish's 25 BANNED Epsom Salt Hacks from the 1950s That Doctors Now Say Were GENIUS
 The Korean Bucket That Grows Unlimited Plant Steroids -- Doubles Every 48 Hours
The Korean Bucket That Grows Unlimited Plant Steroids -- Doubles Every 48 Hours
 This CHICKEN Lays 365 EGGS Per Year. Never Gets Sick. Why Did They Make It Disappear?
This CHICKEN Lays 365 EGGS Per Year. Never Gets Sick. Why Did They Make It Disappear?
Top Tech News
 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...
 Our Emergency Water Plan Wasn't Good Enough - So We Built This
Our Emergency Water Plan Wasn't Good Enough - So We Built This
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
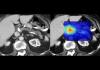 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Microwave Water Splitting for Breakthroughs for Making Hydrogen, Oxygen and Fast Battery
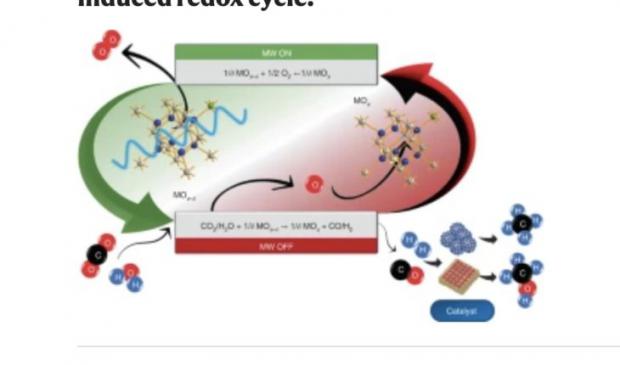
Researchers from the Polytechnic University of Valencia and the Spanish National Research Council (CSIC) report contactless H2 production via water electrolysis mediated by the microwave-triggered redox activation of solid-state ionic materials at low temperatures (less than 250 °C).
This will simplify and significantly cheapen the process for hydrogen production.
Water was reduced via reaction with non-equilibrium gadolinium-doped CeO2 that was previously in situ electrochemically deoxygenated by the sole application of microwaves. The microwave-driven reduction was identified by an instantaneous electrical conductivity rise and O2 release. This process was cyclable, whereas H2 yield and energy efficiency were material- and power-dependent. Deoxygenation of low-energy molecules (H2O or CO2) led to the formation of energy carriers and enabled CH4 production when integrated with a Sabatier reactor. This method could be extended to other reactions such as intensified hydrocarbons synthesis or oxidation.



