
Breaking News
 What if the next billion-dollar dental company isn't making implants...but making them obsolete?
What if the next billion-dollar dental company isn't making implants...but making them obsolete?
 Yemen's Houthis Declare Red Sea Ban on Israeli Vessels,...
Yemen's Houthis Declare Red Sea Ban on Israeli Vessels,...
 The Ogallala Aquifer Is Dying - As The Largest Supply Of Groundwater In The United States Vanishes,
The Ogallala Aquifer Is Dying - As The Largest Supply Of Groundwater In The United States Vanishes,
 We Should Not 'Integrate' Our Military with Any Foreign Nation!
We Should Not 'Integrate' Our Military with Any Foreign Nation!
Top Tech News
 World's longest-range airliner takes to the skies
World's longest-range airliner takes to the skies
 Batteries That Use Sodium Instead of Lithium Could Be Low-Cost Rival to Tesla's
Batteries That Use Sodium Instead of Lithium Could Be Low-Cost Rival to Tesla's
 Elon and SpaceX Have Made AI Training 10 Times Faster
Elon and SpaceX Have Made AI Training 10 Times Faster
 Oklo COO Says Nuclear Waste Could Power America For 150 Years
Oklo COO Says Nuclear Waste Could Power America For 150 Years
 SpaceX Announces LARGEST Starship Mission Ever! They've never done this before!
SpaceX Announces LARGEST Starship Mission Ever! They've never done this before!
 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...
 Our Emergency Water Plan Wasn't Good Enough - So We Built This
Our Emergency Water Plan Wasn't Good Enough - So We Built This
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Precise control of the properties of plastics
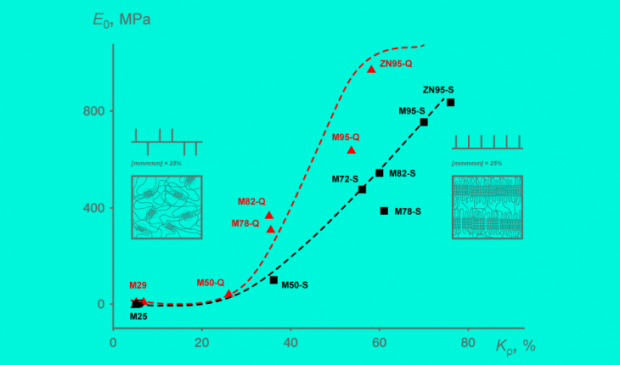
Their new insights make it possible to synthesize a material with predetermined properties, such as elasticity or hardness.
Polypropylene is so ubiquitous one might call it the king of plastics. In terms of production volume, it is second only to polyethylene. By tweaking its molecular structure, polypropylene can be used to manufacture materials with a wide range of features, from elastic bands to high-impact plastic. However, the relationship between the polymer's chemical structure and its mechanical properties was not fully understood.
A polypropylene chain consists of a backbone of carbon atoms with attached hydrogen atoms. Every other carbon atom in the chain has a methyl group attached to it. Two adjacent carbon atoms in the chain with the hydrogen atoms and the methyl group bonded to them constitute a repeating unit called propylene, or propene. The spatial configuration of the macromolecule — the polymer chain — is determined by the mutual orientation of the methyl groups in the chain : If they are all on one side, the molecule is said to be isotactic.

 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater

