
Breaking News
 Palantir Manifesto Shows The Clear Convergence Of Technofascism With Technocracy
Palantir Manifesto Shows The Clear Convergence Of Technofascism With Technocracy
 Washington's Democrat ex-governor says she's disgusted at millionaires' tax...
Washington's Democrat ex-governor says she's disgusted at millionaires' tax...
 The Odyssey Backlash Goes NUCLEAR - WTF Nolan?
The Odyssey Backlash Goes NUCLEAR - WTF Nolan?
 He Got Banned From Selling Skateboards | Joe Rogan
He Got Banned From Selling Skateboards | Joe Rogan
Top Tech News
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
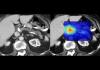 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
A multi-terrain robot from China is going viral, not because of raw speed or power...
 The World's Biggest Fusion Reactor Just Hit A Milestone
The World's Biggest Fusion Reactor Just Hit A Milestone
 Wow. Researchers just built an AI that can control your body...
Wow. Researchers just built an AI that can control your body...
 Google Chrome silently installs a 4 GB AI model on your device without consent
Google Chrome silently installs a 4 GB AI model on your device without consent
 The $5 Battery That Never Dies - Edison Buried This 100 Years Ago
The $5 Battery That Never Dies - Edison Buried This 100 Years Ago
Tissue engineering, replacement organs and regenerative medicine are getting friendlier regulations
The FDA intends to promote the "least burdensome" rules for companies big and small that are seeking to develop new therapies, "while ensuring patient safety."
"Our policy will allow product manufacturers that time to engage with the FDA to determine if they need to submit a marketing authorization application and, if so, seek guidance on how to submit their application to the FDA for approval," Gottlieb said.
The new rules are in keeping with provisions from the 21st Century Cures Act, passed by Congress in December. That legislation earmarked $6.3 billion in funding, mostly for the U.S. National Institutes of Health, towards groundbreaking medical research.
Over the past few years scientists and physicians have developed tissue-engineered skin for transplant; bladders grown from a patient's own cells; and tissues grown to repair ailing hearts or failing knees.



