
Breaking News
 Economist Issues a Chilling Warning (You Should Prepare)
Economist Issues a Chilling Warning (You Should Prepare)
 The Internet Is Getting Harder to Trust | Josh Smith From #485 | The Way I Heard It
The Internet Is Getting Harder to Trust | Josh Smith From #485 | The Way I Heard It
 DIY LFP Battery Explosion! Is it Possible??
DIY LFP Battery Explosion! Is it Possible??
 House Leadership Delays Vote on Ending the Iran War
House Leadership Delays Vote on Ending the Iran War
Top Tech News
 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...
 Our Emergency Water Plan Wasn't Good Enough - So We Built This
Our Emergency Water Plan Wasn't Good Enough - So We Built This
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
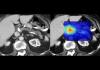 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
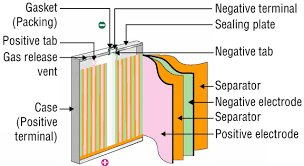
Lithium-ion battery boost could come from "caging" silicon in graphene
The researchers managed to remove two long-standing barriers to these improvements by putting silicon particles in graphene "cages."
To improve capacity in recent years batteries have begun to use silicon anodes, which have more capacity than the graphite conventionally used. But silicon particles also swell so much during charging that they're prone to cracking or shattering and they can also react with the battery electrolyte, forming a coating that reduces performance.
The solution from the team at Stanford and the Department of Energy's SLAC National Accelerator Laboratory is to encase each silicon particle in a "custom-fit cage" of graphene. At only one-atom thick, graphene is the thinnest, strongest form of carbon and also conducts electricity well.
The carbon cages would allow the silicon to expand and even break apart, but keep the pieces together so that they can continue to function. The graphene barrier would also block the destructive chemical reactions with the electrolyte from occurring.



