
Breaking News
 Martin Armstrong Warns the Financial World Order Is Breaking Apart | Part 1
Martin Armstrong Warns the Financial World Order Is Breaking Apart | Part 1
 Thank You Veterans – Memorial Day Remembrance
Thank You Veterans – Memorial Day Remembrance
 Interview: No One is Talking About This and its BIGGER than HORMUZ
Interview: No One is Talking About This and its BIGGER than HORMUZ
Top Tech News
 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...
 Our Emergency Water Plan Wasn't Good Enough - So We Built This
Our Emergency Water Plan Wasn't Good Enough - So We Built This
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
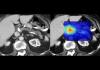 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Chemists have successfully produced fuels using water, CO2 and visible light through artificial phot
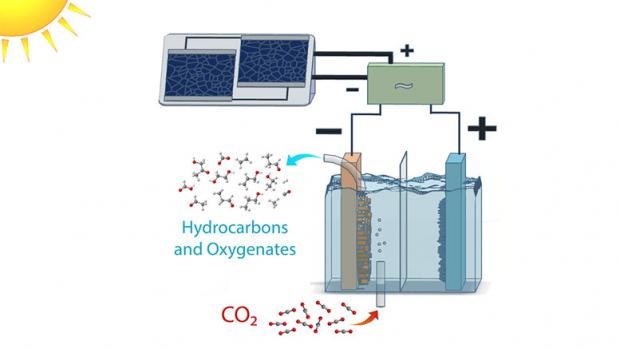
Photochemical conversion of CO2 into fuels has promise as a strategy for storage of intermittentsolar energy in the form of chemical bonds. However, higher-energy-value hydrocarbonsare rarely produced by this strategy, because of kinetic challenges. Here we demonstrate a strategy for green-light-driven synthesis of C1-C3 hydrocarbons from CO2 and H2O. In this approach, plasmonic excitation of Au nanoparticles produces a charge-rich environment at the nanoparticle/solution interface conducive for CO2 activation, while an ionic liquid stabilizes charged intermediates formed at this interface, facilitating multistep reduction and C-C coupling. Methane, ethylene, acetylene, propane, and propene are photosynthesized with a C2+ selectivity of ~50% under the most optimal conditions. Hydrocarbon turnover exhibits a volcano relationship as a function of the ionic liquid concentration, the kinetic analysis of which coupled with density functional theory simulationsprovides mechanis

 Massie Interview on War
Massie Interview on War


